Blue ice packs, also know as gel ice packs, are able to get extremely cold without completely freezing solid. This makes the very useful for applying to injuries on your body where you need to mold the ice pack over the skin.
They are also commonly used in lunch boxes to keep everything cold.
But what is exactly in these things and is the liquid in ice packs toxic? What would happen if you were to accidentally swallow it? Could it potentially kill you?
Generally speaking, the ingredients used to make ice gel packs are non-toxic and can contact the skin or be consumed in small amounts without major issues. The chance of being poisoned by a gel ice pack is extremely low and you would need to consume large quantities to experience health issues.
While that is the main answer I wasn't satisfied with that and I wanted to do some digging of my own to find out what ingredients are actually used in ice packs and what kind of health threat does each of these ingredients pose if the ice pack was to break and get on our skin or if we swallowed some?
What Are Ice Packs Made From And Are They Toxic?

The major premise behind an ice pack is simple – be able to put it in the freezer at 0° F (-18° C) and have it get slushy but not frozen solid.
This can be achieved using a variety of different methods and a variety of different ingredients – some more toxic than others.
Below we will look through the major and most common gel types used in ice packs and explore how toxic they are.
I will put a disclaimer out there that I am NOT a chemist. I am simply exploring this as someone who uses these products and wants to know more about them.
It's actually really hard to find what ingredients are used in gel packs as they don't generally list their ingredients but this article was a great help in uncovering the gelling agents commonly used.
Water
The major ingredient in almost every single ice pack or gel ice pack is going to be water.
I think it goes without saying that water is non-toxic by itself and can be consumed in large quantities without issue.
Hydroxyethyl Cellulose
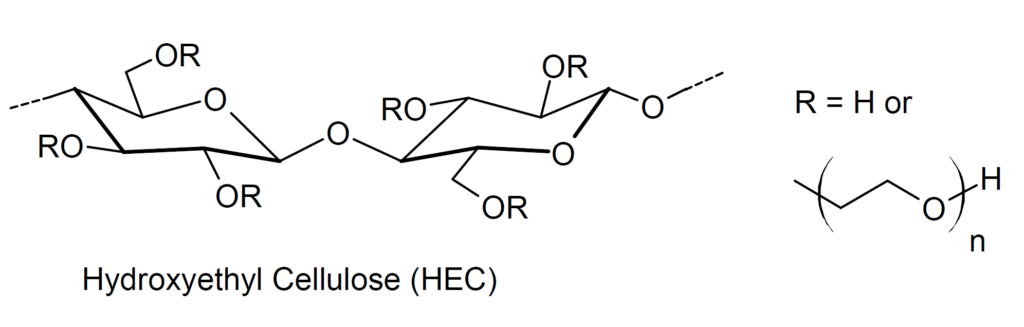
This gel/thickening agent is NON-TOXIC and is unlikely to harm you if it is consumed or if it comes in contact with your skin
According to Wikipedia:
Hydroxyethyl cellulose is a gelling and thickening agent derived from cellulose. It is widely used in cosmetics, cleaning solutions, and other household products.
It's also used in KY Jelly, which is lubrication obviously intended to be put on your skin and also consumed.
There is also this 2-year study on rats where they consumed Hydroxyethyl Cellulose. The summary states:
Indirect evidence, based on feeding experiments to rats during a two‐year period, indicates that hydroxyethyl cellulose is neither absorbed or hydrolyzed in the gasrrointestinal tract.
Propylene Glycol

Propylene glycol helps gives ice packs their thick gel like feel and lowers the freezing temperature.
Propylene glycol is considered to be low toxicity. You would have to consume large quantities to cause health effects in humans, it's essentially non-irritating to the skin and minimally irritating to the eyes.
Wikipedia has a large article on propylene glycol and its uses. It's often used in antifreeze and is also used in some types of foods as well as electronic cigarettes.
Propylene Glycol is another thickening agent
Silica Gel

Silica gel beads can sometimes be used in gel packs. Because they absorb water well and don't stick together they can be frozen but still be able to be molded onto unusual shapes.
Silica gel is non-toxic and chemically un-reactive. According to this article it is basically sand and is not harmful if swallowed.
SAP (Super Absorbant Polymer)
Super absorbant polymer (SAP) is the absorbant gel crystals used in things like diapers to stop baby's wee leaking everywhere. They can also occasionally be used in ice packs.
The risk with SAPs is where the total expanded size of the SAP is larger than the diameter of your small intestine. This is about 25-30mm or about 1 inch in a small child.
A dry SAP bead of about 8 mm or less is generally considered safe and given that SAPS used in ice packs aren't dry and generally aren't larger than 1 inch in diameter they shouldn't pose any major health risks.
Isopropyl (Rubbing) Alcohol

A common method for making reusable ice packs at home is to combine water with isopropyl alcohol, also known as rubbing alcohol.
This is the cleaning alcohol you can buy from your supermarket and it's very affordable. It has a low freezing temperature and at a ratio of 2:1 or 4:1 water to alcohol it forms a slushy consistency when put in a freezer.
Of all the common ingredients used to make gel packs this is the most dangerous. Consuming it can lead to drunkenness, headaches, dizziness, nausea, vomiting, low blood pressure, comas and even death!
It is suggest that 8 oz of this type of alcohol is enough to kill you (according to this article) if you don't seek medical attention.
Given a ratio of 2:1 water to alcohol or 4:1 you would need to consume somewhere between 24 oz (710 mL) to. 40 oz (1.2 L) in order to consume a lethal dose.
In small children the quantities would be less.
Salt Water

You can also make a reusable ice pack with salt water. It is advised you use around a 12:1 ratio for water to salt when making an ice pack.
Salt is considered non-toxic in small amounts. According to Gizmodo you would need to consume around 47 teaspoons or about 8 oz of salt in order to cause serious harm.
At a 12:1 ratio this means you would need to consume 104 oz (3.1 L) of a salt water ice pack in order to cause serious hard.
Is The Blue Liquid In Ice Packs Toxic To Touch?
Ice packs are generally kept in thick sealed plastic bags so none of it is exposed to your skin.
It often also has a weird goey like texture and given you don't know what it's it you may wonder if it is toxic to touch.
The blue liquid in ice packs is considered non-toxic and can come in contact with your skin without any major issues. Most ingredients used are considered to be non-irritating to skin and only mildly irritating to the eyes. It is advised you wash it off quickly if contact does occur.
Of all the ingredients we mentioned up above the isopropyl alcohol (IPA) is the most dangerous as it can be absorbed into the body through the skin.
However, small amounts of IPA on the skin shouldn't cause any major issues but large and extended exposure can cause isopropyl alcohol poisoning and medical attention may be required.
Given than ice packs generally dilute IPA with water at a ratio of 2:1 or 4:1 water to alcohol the risk is lowered dramatically when compared to exposure of pure IPA.
If you do come in contact with IPA from an ice pack this article advises:
When contact with the skin has occurred, the affected skin should be flooded with water and then gently and thoroughly washed with soap and water. In the event of a large spill in contact with skin, wash and monitor for signs of poisoning, seek medical treatment if symptoms appear.




